Author's posts
Nov 15 2009
The past is a foreign country
I’ve been hanging out with historians recently (both online and in real life), so it got me thinking about how scientists treat history. The 150th anniversary of the publication of “On the Origin of Species” is coming up too, so it seemed like a good time to write this post.
My impression is that historians are about the reading of contemporary material, and drawing conclusions from that material; a realisation I came to writing this is that historians seem to have the same sense of wonder and passion for historical minutiae as I have for nature and science. I remember talking to a historian of science who was working on an original manuscript of some important scientific work, it quickly become clear that this was much more exciting for her than me. To me the exciting thing was the theory presented in it’s modern form, I wasn’t very interested in the original.
In science it isn’t the original presentation that’s important: I haven’t read Newton’s Philosophiæ Naturalis Principia Mathematica, Maxwell’s A Treatise on Electricity and Magnetism, any of Einstein’s four “Annus Mirabilis” papers, Galileo’s Dialogue Concerning the Two Chief World Systems, Darwin’s On the Origin of Species, the list goes on…
And that’s not to mention the real contemporary material: correspondence, notes and labbooks. I have a sequence of about 20 labbooks in the loft from 15 years of research, supplemented by a hoard of files and e-mails stored on my computer, covering the same period. I’m not sure I even want to try to reconstruct what I was thinking over that period – let alone try it on someone else’s records! It’s not that I’m remiss as a scientist, we just don’t read original material.
The original presentation of an idea may not be the clearest, and it may well be that it makes more sense later to present it as part of a larger whole, and to be honest scientists can be a bit hit and miss: Newton’s physics is great but the alchemy was bonkers. Science comes in bits, these days the bits are the size of a journal article and it’s only when you’re doing active research at the cutting edge that you need to keep track of the bits.
Mathematical notation is an issue for original publications. For example, Maxwell’s equations, which describe electromagnetism (radio waves, electricity, light…) are a monster in his original presentation but can be squished down to four short lines in modern notation (actually a notation introduced not long after his original paper). There’s a rule of thumb that each equation in an article halves the number of readers, therefore I link you to Maxwell’s 1865 version on page 2 of this document with the modern version at the bottom of page 6…
impressive, no?
A bit of history is introduced into the teaching of science but it’s either anecdotal such as the apple falling on Newton’s head, Gallileo dropping things off towers, Sadi Carnot and his wacky exercises, or we might give a quick historical recap as we introduce a subject. But to be honest it’s really all window dressing, the function of this history is to provide a little colour and give students the opportunity to do some exercises which are tractible.
Are scientists losing out as a result of this historical blindness? History should certainly inform us of our place in society, and our future place in society (okay – I’m talking about cash here!). I’m less sure that it has something to teach us on the ‘craft’ of science, this is something that comes from professional training – perhaps it would help if we were not presented with such caricatures of our scientific heroes.
So that’s my view, how wrong can I be?
Nov 09 2009
Pretty molecular models
And now I leap off into a topic in which I am not properly trained: molecular biology!
You sometimes get the impression that scientists lead dull lives because they over-analyse things, they’ve lost their sense of wonder. The thing is: the more you know, the more you wonder.
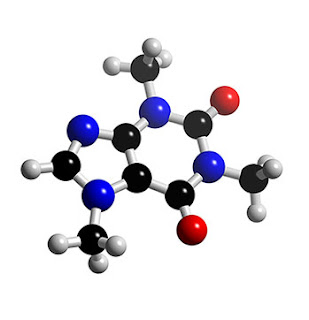
One step up from atoms, you find molecules – atoms bound together. Starting things simple, here’s caffeine:
As every chemist kno carbon (C) atoms are black, nitrogen(N) atoms are blue, oxygen(O) atoms are red and hydrogen (H) atoms are white. (Not really but those are their traditional colours in molecular models). Isn’t it beautiful? You can play with an interactive version here. In real life chemistry is more messy than this which is why I’m a physicist rather than a chemist.
The caffeine molecule is about 1 nanometer across, 1 (US) billionth of a meter. To give you a feel for the size of a nanometre: think of a grain of rice – about 1mm across, now imagine a kilometre. Walk your kilometre with the grain of rice, I walk a kilometre in about ten minutes and it takes me past two roundabouts, a gym and a postbox. Now look at you grain of rice again. To a caffeine molecule, a grain of rice is a kilometre wide.
Molecular models of this sort are a representation of reality, the things they miss out are: (1) in real life molecules are not static, they’re jiggling away furiously through the action of thermal energy (2) generally they’re going to be surrounded by solvent molecules (often water, which are also zipping and wiggling around) (3) they’re sort of soft, fuzzy and deformable and different parts of the molecule will be sticky or slippery according to their chemical nature. Ten years ago a good question at any molecular modelling seminar was to ask about the solvent molecules, the usual answer was “there aren’t any” – this usefully puts molecular modellers in their place since we’re rarely interested in molecules without solvent. Perhaps things have moved on since those days.
Life specialises in bigger molecules than caffeine, exquisitely crafted into little machines. And the incredible things is that all of life (humans, mammals, reptiles, birds, snails, bees, tardigrades, sponges, plants, algae, bacteria, fungi, weird bacteria that live in hot underwater vents) share the same 4-letter DNA code, which codes for the same set of 21 amino acids which build all the proteins to make life. Many of the proteins themselves aren’t hugely dissimilar across all the plant and animal kingdoms, particularly those to do with the most basic operations (processing DNA, converting food to energy).
Proteins are strings of amino acids: each different type of protein has a different sequence of amino acids.
Protein molecules typically contain many (a hundred or more) amino acids. The amino acid sequence is known as the primary structure, next up is the secondary structure: alpha-helices and beta-sheets. Different amino acid sequences can produce alpha-helices and beta-sheets that look the same. These structures are represented using “ribbons”:
This is a model of lysozyme, the alpha-helices are shown in red and the beta-sheets are yellow, bits of “random coil” amino acid sequence are shown in green. Lysozyme is about 5 nanometres from one end to the other. You can play with an interactive version here. The amazing thing about proteins is that their 3D structure forms spontaneously and very rapidly when they are synthesised in the cell, this process is known as ‘folding’. Furthermore the folded, or tertiary structure, of the protein is the same every time – it has to be or the protein won’t do it’s job. One of the great challenges in molecular biology is that, despite knowing the amino acid sequence of a protein from the DNA which encodes it, working out the 3D structure is a question of measurement, or comparison with other sequences of known folded structure.
Lysozyme is a physicist’s protein, you can buy it in bottles by the gram. I’ve worked on lysozyme, looking to see how it unfolds on a surface when heated.
You can go see more protein structures on http://proteopedia.org/, the lysozyme model above is 132L. I could play on there for hours…
References
Green, R.J., Hopkinson, I. & Jones, R.A.L. Unfolding and intermolecular association in globular proteins adsorbed at interfaces. Langmuir 15, (1999), 5102-5110.
Oct 31 2009
Bryn Alyn – an autumn walk
Off to Llanferres yesterday for an autumn walk through the woods, along the ridge and back again. You can see the route here:
View Bryn Alyn in a larger map
It’s a variant on a route in “Walking in the Clwydian Range” by Carl Rogers. A tiny domestic detail: whilst walking the Inelegant Gardener carries the guide book, preferring to navigate via prose and I carry the OS map – preferring maps. The track is from a Garmin GPS60, I don’t use it for navigation but to tag my photos with location information for which I’ve written a little program.
We last did this walk in May, but I committed a terrible faux pas: the battery on my camera went flat and for some reason I’d not brought a spare and had deliberately left behind my second camera and my phone (which also has a camera, which is really crap).
Beech trees were definitely the best for autumnal colours, although birch produces an attractive pointillist effect, sycamore seemed best for kicking through.
At the top of the initial climb there is a little bit of limestone pavement, this is most famously found above Malham Cove but it’s nice to find your own little patch.
For reasons I can’t explain I like the stray bits of ironwork left over from old fencing, parts of the Lake District are particularly good for this.
And to top it all off, a cow wearing a ginger wig:
This was one of many cows in a field we passed through, they were fine looking beef cows in a wide range of colours. We also had a “That’s no cow, it’s a bull” moment but I was reassured by remembering vaguely someone saying that you’re okay in a field with a load of cows and a bull because the last thing on the bull’s mind is going to be you. If this isn’t actually true then I’d prefer to be left in ignorance, if you don’t mind.